Abstract
Our client wanted us to explore how technology could help reduce patient and site burden to participating in clinical trials.
Over the course of this nine-week project, our team researched challenges and opportunities within the current clinical trial model, presented an ideal-state model for how the company could sponsor distributed trials in the future, and developed a pitch deck and strategy for leadership to connect this ideal state with ongoing initiatives within the company and build momentum for change.
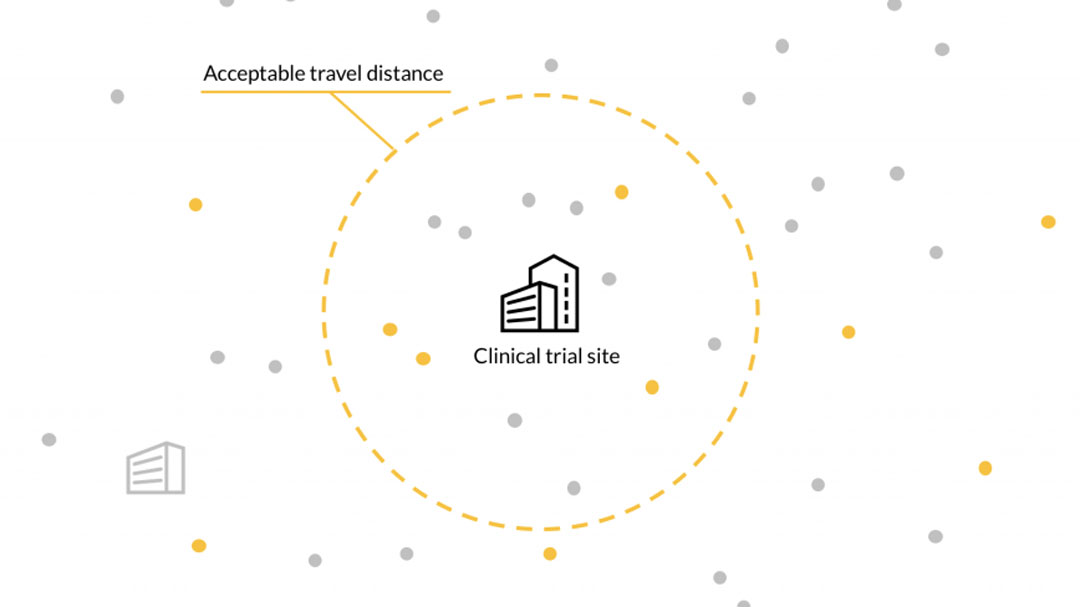
Challenge
The current clinical trial system presents major logistical challenges for patients, sites, and sponsors. Patients who could benefit from clinical trials are often not offered the option, and trial requirements often impose a significant time and travel burden for those who do participate. Increasingly stringent trial eligibility criteria also means it is becoming much more difficult for sites to find and enroll patients.
Approach
Our team reviewed the current clinical trial ecosystem and identified opportunities and technology offerings with the potential to reduce site and patient burden.
Based on this review, we proposed an ideal-state trial model, and prepared a pitch presentation our client could use to build organizational buy-in for the ideal-state model.
Outcomes
A key finding was the importance of involving patients and sites in protocol design; though new technologies could improve communication with patients, ease trial management, and improve regulatory compliance, even the best technology advances can’t overcome a trial protocol design that isn’t responsive to patient and site needs.
Our client is using this research to inform internal process changes, initiatives, and partnerships to make their trials more patient-centric.
Project excerpts
First, identify challenges and opportunities that could be addressed though a distributed clinical trial model.
Our team’s user researcher spoke with stakeholders, external experts, patients, and providers to hear about challenges within the current system and their reactions to the distributed clinical trial model. The project lead and I conducted a literature review to understand barriers to patient access and engagement with clinical trials more broadly.
Deliverables
- Literature review
- Patient and provider interviews
- Interview findings report
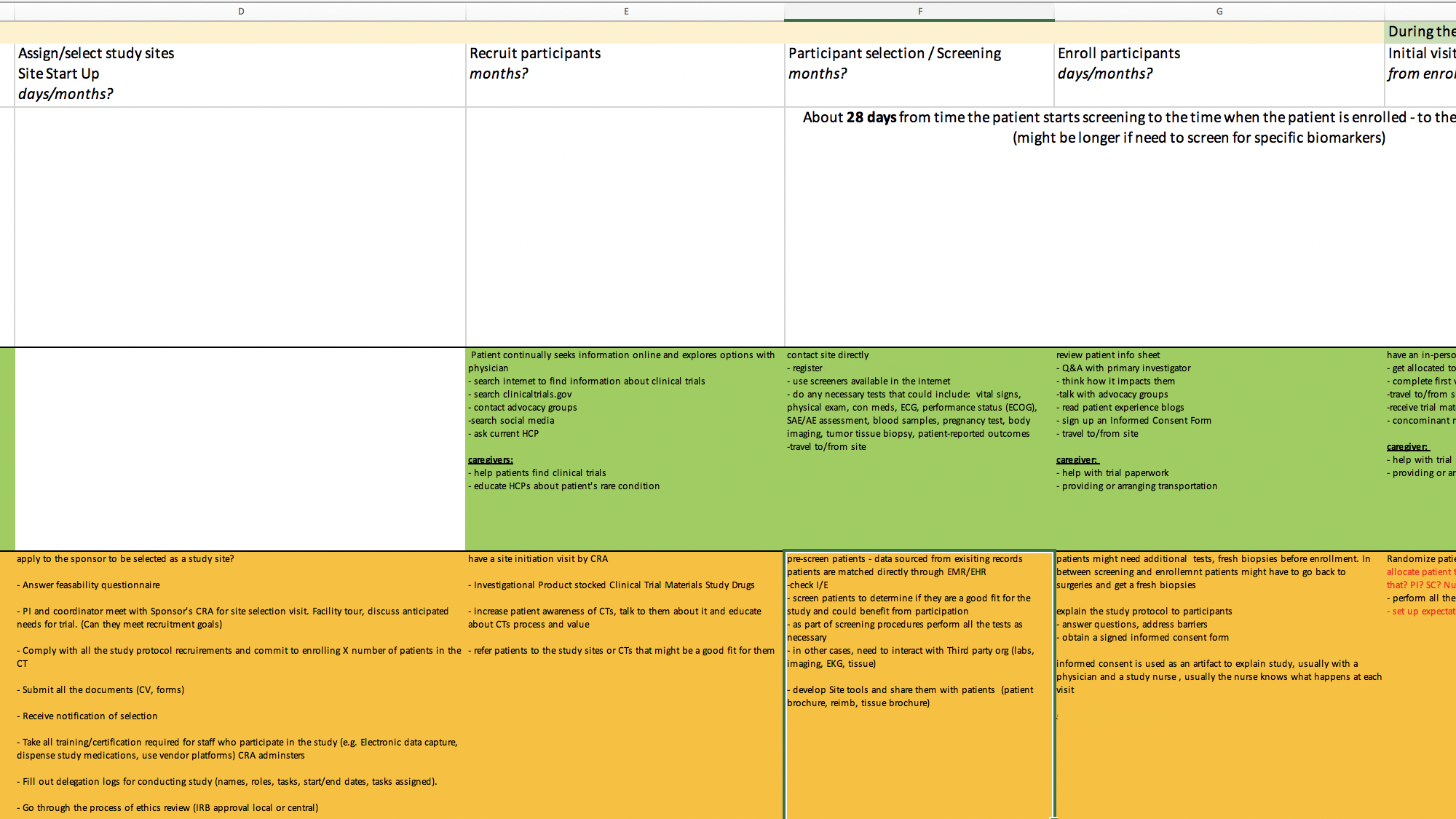
Excerpt from our clinical trial process spreadsheet
Next, synthesize findings into a current-state map and work with our client to prioritize opportunities.
Once we understood the current state of clinical trials and key challenges, the project lead and I identified opportunities at each trial stage. We worked with our team’s visual designer to develop an ecosystem map to visually represent trial burden at each stage for patients, sites, and sponsors and highlight areas of overlap.
The project lead and I used these references to facilitate a workshop with our clients and identify the highest value opportunities. Together, we selected the most promising strategies to incorporate into our final vision for an ideal-state distributed trial model.
Deliverables:
- Overview of opportunities
- Design workshop
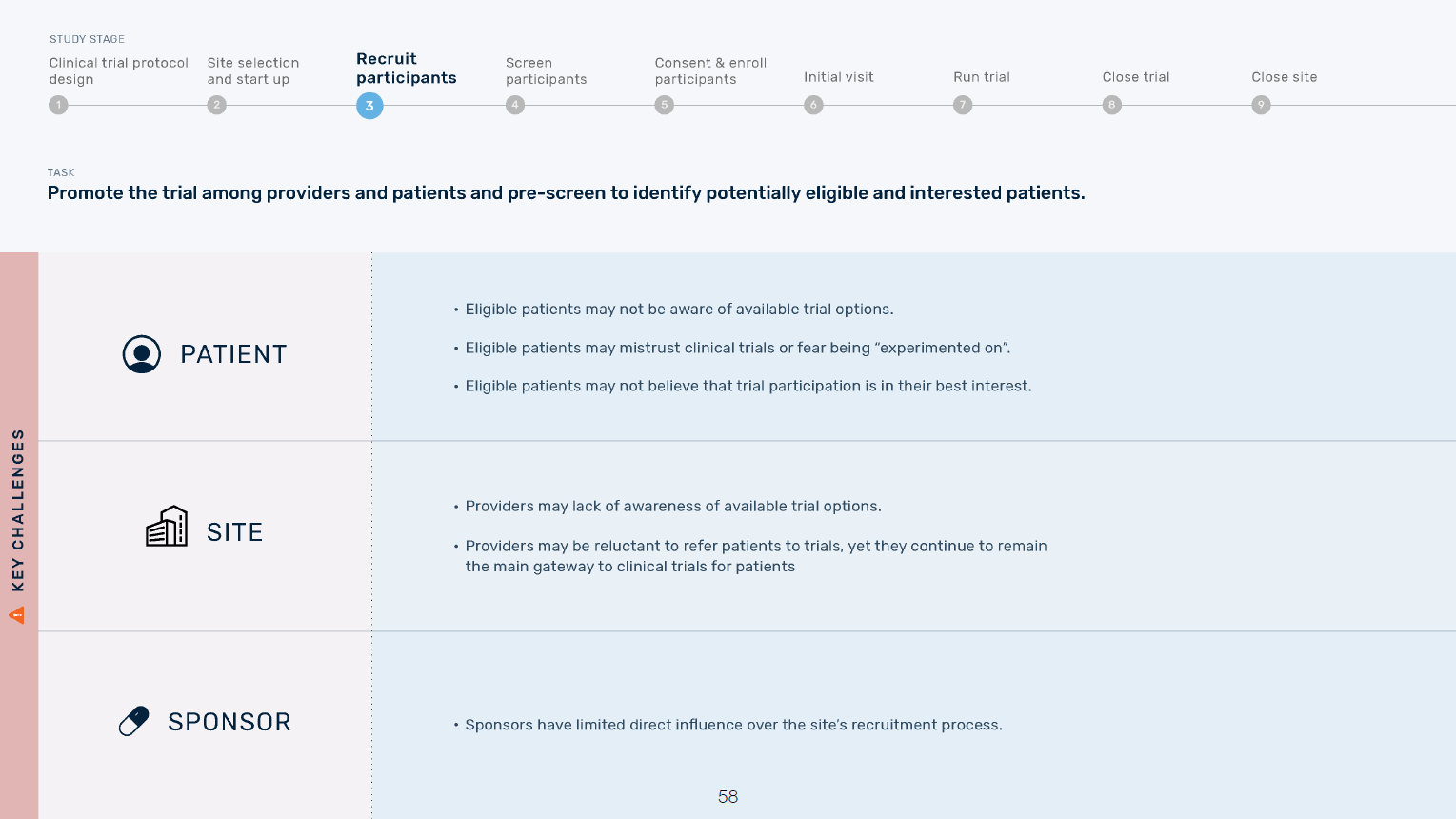
Excerpt from overview of patient, site, and sponsor challenges at each trial stage
Finally, present an ideal-state distributed trials model to pitch to leadership.
We took the insights from our workshop and developed an ideal-state map of clinical trials that highlighted current initiatives and how they fit into the broader vision for improving patient access. The project lead and I worked together to distill over 100 pages of findings, reports, and diagrams into a 15 minute presentation that our clients could use to win internal support for the initiative.
Deliverables:
- Ideal-state distributed clinical trial ecosystem map and patient journey map
- High-level strategy presentation for our clients to gain internal support for the initiative

Pitch presentation excerpt
Team contribution
I helped conduct and synthesize information from the literature review and interviews and map insights to each clinical trial stage. The project lead and I worked together to develop recommendations, map the clinical trial system for our visual designer to further refine, and develop a strategy for our client to pilot our recommendations. I was responsible for creating and delivering the final strategy presentation to our client. My thanks to our team’s user experience researcher for conducting provider and patient interviews, and to our team’s visual designer for creating final graphics and diagrams.